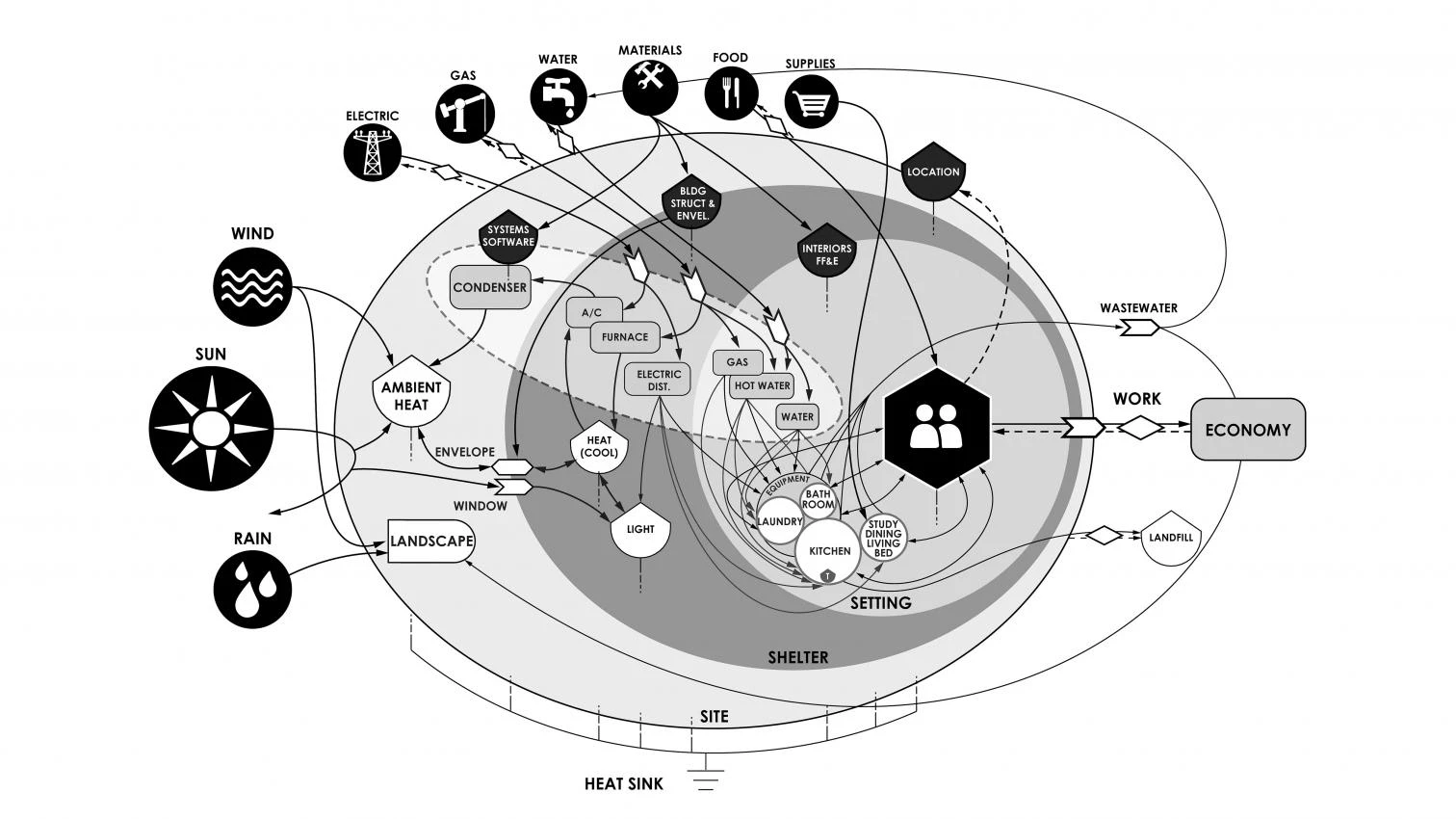
Thermodynamics has largely been implemented with the measure of efficiency, and it has been one of the fundamental tools of technological innovation. Reviewing the different uses of efficiency helps reveal the difference between the different principles of thermodynamics. Count Rumford observed the heat equivalence of work in 1798, based on his measurements of the frictional heat released while boring cannons. The theory that forms the basis of the first law was not accepted until the 1840s, when Joule and Mayer independently showed the same correspondence (with more precision) in the interactions between mechanical work, electricity, magnetism, chemical reactions (combustion and digestion), and heat. The energy defined in the first law is ultimately the abstract quantity that is conserved in all those interactions, which is measured as heat. Efficiency measurements of the first law are therefore expressed in heat equivalents, comparing, for example, the heat content of the fuel burned in a machine to the heat value of the work delivered at the end...





